e-mail:
office@ukrain.ua

vul. Kopernyka 26/5,
79000 Lviv, Ukraine
Tel./Fax: +38 032 2611652
|
УКРАЇНСЬКОЮ | |
| “UKRAIN COULD REPLACE CHEMOTHERAPY IN TREATING ALMOST ALL CANCERS” | ||
| Robert C. Atkins, M.D. |
||
| Nowicky Pharma e-mail: office@ukrain.ua |
|
|
Cancer can be reversed |
CANCER CAN BE
|
||
There is a plant
modified alkaloid |
 |
Greater celandine contains more than 40 alkaloids. For Ukrain some modified alkaloids are used which are relevant for cancer treatment. |
UKRAIN |
||
- treats cancer
|
||
|
|
UKRAIN IS REGISTERED IN MANY COUNTRIES: |
|
UKRAINE
GEORGIA
UNITED ARAB EMIRATES
MEXICO
TUNISIA |
AZERBAIJAN TAJIKISTAN TURKMENISTAN 1995 - #1330-95 |
USA AUSTRALIA
|
||
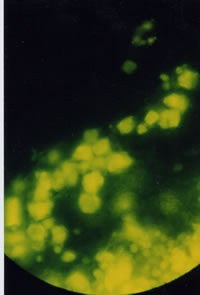 |
Ukrain autofluorescence in UV-light. A 68 year old female patient was diagnosed as having inoperable pancreatic head carcinoma complicated with jaundice. Ukrain was administered intravenously at a single dose of 20 mg 4 hours before palliative surgery. During surgery, fine needle biopsy of the pancreas was performed. Auto-fluorescence of Ukrain under UV-light is seen in cancer cells only. | |
THERAPY SUCCESSES WITH UKRAIN: |
||
- more than 70% complete remissions with prostate cancer - some patients diagnosed with metastasising breast cancer have lived without recurrence for more than 25 years thanks to Ukrain as monotherapy - some patients diagnosed with metastasising colonic cancer have lived without recurrence for more than 20 years thanks to Ukrain as monotherapy - the best therapy for children with cancer because the therapeutic dose of Ukrain has no side-effects
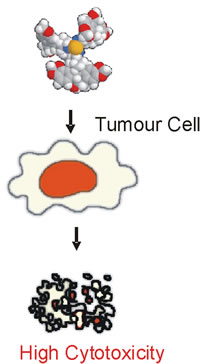
- destroys only cancer cells while leaving healthy cells undamaged |
||
 |
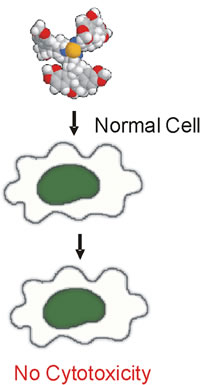 |
This has been proved by research experiments on more than 100 cancer cell lines and 9 normal cell lines. National Cancer Institute, Bethesda, Maryland, USA |
- regenerates the immune system
|
||
 |
Pancreatic adenocarcinoma: pre-operative treatment with Ukrain. A total of 100 mg 10 days before the operation. Formation of a capsule (A) around the tumour. The cancer cells do not penetrate this capsule. | |
| Massive infiltration of round cells (B) on the periphery of the tumour capsule (magnification, x100). | ||
Understanding, as well as possible modulation, of this mechanism involved in selective cancer cell death could be used as a model for providing safe and efficacious new therapies for cancers.University of Rochester Cancer Center
|
…Ukrain is capable of improving the general conditions and prolonging the lives of terminal cancer patients by boosting their immune systems and inhibiting tumour growth.
|
|
Ukrain is a unique product that acts in at least two ways on cancer (by killing cells and by increasing the immune response).Ralph W. Moss, Ph.D.
|
Our results suggest Ukrain as a potent drug for the treatment of unresectable pancreatic cancer.Prof. H.G. Beger et al., University of Ulm,
|
 |
 |
 |
 |
 |
 |
 |
 |
17.11.2010 |
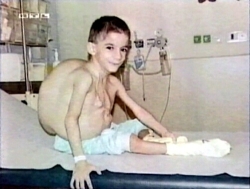
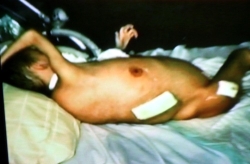
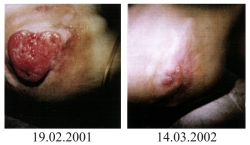
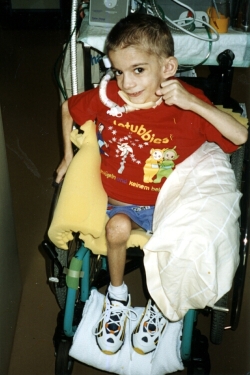
| The sad story of Stefan Dan is known to many people from the book Buch „Krebsmittel Ukrain - Kriminalgeschichte einer Verhinderung“ von Dr. Eleonore Thun-Hohenstein (Molden Verlag, Vienna 2004), many press reports, (Link 1, Link 2, Link 3) from television ("Die Redaktion spezial", RTL2; "Mona Lisa", ZDF, 10 October 2004, 18:00, http://www.presseportal.de/meldung/604528) as well as from the specialist literature. (Link) |
More than 40 alkaloids have so far been detected in greater celandine. Each alkaloid has its specific effect on the human body. Only those alkaloids which are relevant for cancer treatment are used in UKRAIN. UKRAIN accumulates very quickly in cancer tissue. It is the first product which only attacks cancer cells while leaving healthy cells undamaged. |
As is known from the Internet around 10.8 million people are diagnosed with cancer every year and approximately 6.7 million of them die. Thus, despite all the medicines available, only 38% can be saved. Much time is required before a medicament comes on the market, which is why the World Health Organisation has also permitted doctors to prescribe non-approved preparations to save the lives of patients with this disease. On the basis of this provision Austria has § 8.(1)2 Arzneimittelgesetz (Medicines Act), which we quote as follows: "Medical specialities do not require approval if a doctor, dentist or veterinary surgeon licensed to practice in Austria attests that the medical speciality is urgently needed to combat a life-threatening situation or serious damage to health and that this result could not be expected to be achieved with an approved and available medical speciality according to the state-of-the-art." There is an almost identical law in every country (e.g. in Germany § 73.3 AMG). Ukrain is not approved in Austria but in many other countries. Research findings on Ukrain have appeared in 262 specialist publications and been presented at 294 international medical congresses. Its efficacy, safety and quality have been confirmed by 260 scientists from 24 countries at 60 universities and research institutes. Thereby, every doctor can also prescribe Ukrain under the conditions described above. |
| UKRAIN AND ITS POTENTIAL |
Another example is a case of a colon cancer patient whose situation after surgery in 1988 has been evaluated as very serious and the prognosis was poor (case colon cancer) A leading Vienna oncologist evaluated this case as follows: ‘There is a status after surgery of an ulcerating colon adenocarcinoma with lymph node metastasing. Noteworthy is a large primary tumour, its ulceration and, moreover, the poor differentiation. All these factors indicate an unfavourable prognosis.’ Mrs. Jakob has been treated with Ukrain as monotherapy and is still alive without recurrence – 20 years after surgery. Ukrain can cause the full regression of the main tumour and also of metastases. In the treatment of advanced tumours Ukrain can improve the quality of life and prolong survival. Many clinical studies have proved this, such as those of the work groups led by Prof. Beger in Germany and of Prof. Zemskov in Ukraine with pancreatic cancer (Gansauge 2002, Gansauge 2003, Gansauge 2007, Zemskov 2002), as well as groups led by Prof. Susak and Prof. Bondar in Ukraine with colon cancer (Susak 1996, Bondar 1998). Neoadjuvant (before surgery) use of Ukrain can induce encapsulation of tumours as revealed the studies by the researchers of Grodno Medical University (Grodno, Belarus) in breast cancer (Uglyanica 1996a, Uglyanica 1996b, Uglyanica 1996c, Uglyanica 1996d, Uglyanica 1996e, Uglyanica 1996f, Uglyanica1998, incapsulation). Ukrain is the first and only anticancer drug which accumulates in cancer cells minutes after administration. In contrast to chemotherapy the drug destroys only cancer cells by inducing apoptosis (programmed cell death) while leaving healthy cells undamaged. At therapeutic dose the product has no appreciable adverse effects and does not damage healthy cells but only attacks cancer cells. Due to its very high therapeutic index of 1250 – in contrast to common cytostatics with a low TI of 1.4-1.8 – there is no danger of an overdose with Ukrain therapy (therapeutic index - ratio of toxic dose to the therapeutic dose). Ukrain also does not cause necroses with intramuscular administration. This drug has been approved as standard anticancer medication in some countries of Europe and Asia (for example, Ukraine: first approval on October 18, 1998, #3641, re-approval on September 2, 2003, #3641; United Arab Emirates: registration no. 4987-6179-1; Georgia, 2008) as well as in Mexico (No. 036M2005 SSA). Ukrain has also been designated as an Orphan Drug for the treatment of pancreatic cancer in the USA (Designation Request #03-1693) and also in Australia (File 004/009839). As noted above, through its antiangiogenic properties this medicine encapsulates tumours, thereby making them accessible to surgery. If possible, it is recommended to reduce the tumour burden within 10-14 days after the start of therapy. The destruction of cancer cells with Ukrain is dose dependent. The higher the dose the more cancer cells are killed. Tumour degradation products such as amino acids, milk acid and others can release and induce transient local pain which can be treated with local sodium hydrogen carbonate compresses or oral intake of small quantities of sodium hydrogen carbonate solution (baking soda on knife top approx. 100-150 mg diluted in 250 ml water). This pain can be also treated with sodium bicarbonate solution as an infusion, see http://www.orthopaedie-dr-woelffle.de/neuraltherapie_neu_ulm.html , http://www.neuraltherapy.com/, http://www.neuraltherapybook.com/. At small doses, this preparation has an immune-modulating effect and significantly enhances malignotoxic activity of macrophages, lymphocytes and NK-cells against various cancer cells, i.e. it has properties related to cell mediated immunity. Due to these immune effects it improves the general condition of patients and is used to treat viral diseases. It is recommended to administer small doses of Ukrain immediately after surgery (during 3-5 days, 5 mg a day). It helps to regenerate the immune function, improve wound healing and rehabilitation and to prevent keloid scarring. For successful treatment large doses of Ukrain should be alternated with small ones: large doses destroy tumours, small doses help to eliminate tumour degradation products. Therefore alternate doses are used in the course of Ukrain therapy, e.g. Monday 5 mg - Thursday 20 mg, or 5 – 30 mg, 5 – 40 mg, twice a week, with a two-day break after a small dose and a three-day break after a large one. There are reports on tumour regression after 10 day course of in-patient Ukrain therapy at a daily dose of 20 mg intravenously. The mechanism of action of Ukrain has been tested on more than 100 cancer cell lines so far, including cisplatin-resistant lines, but also on normal cell lines. Ukrain has been proven to be effective against all cancer lines whereas normal lines were unaffected. In contrast to conventional cytostatics, which are toxic both against cancer and normal cells, Ukrain is toxic only against cancer cells and has been described as a malignotoxic agent. Among other in vitro studies, Ukrain has been tested at the National Cancer Institute, Bethesda, Maryland, USA, on 60 cell lines representing eight important human malignant tumours: brain, ovarian, kidney, small cell and non-small cell lung carcinomas, colon cancer, leukaemia and melanoma. Ukrain was effective against all cell lines (NCI cell lines). In comparison with 5-fluorouracil (5-FU) and gemcitabine, two standard cytostatics in the treatment of abdominal carcinomas, Ukrain achieved better results having caused not only growth inhibition of cell lines but also reduction of cell mass. This advantage of Ukrain over 5-FU and gemcitabine was confirmed with clear clinical benefits in several randomised clinical studies (comparison 5-FU). 96 patients with colorectal carcinomas were included in a randomised study performed at the National Medical University, Kiev, Ukraine. 48 patients were treated with UKRAIN (15 of them with metastasing and 33 with non-metastasing colorectal tumours) and 48 patients were treated with 5-FU and radiotherapy. The survival rate after 21 months was 78.6% in the group treated with UKRAIN and 33.3% in the group treated with 5-FU and radiotherapy (Susak 1996). 48 patients with rectal carcinomas participated in a randomised study carried out at the Regional Cancer Center, Donetsk, Ukraine. 24 patients received high-dosage radiotherapy and 5-FU before surgery, and another 24 patients received one series of UKRAIN therapy before surgery (10 mg on alternate days, up to a total dose of 60 mg) and one series after surgery (up to a total dose of 40 mg). During the following 14 months, 25% of patients who had received 5-FU and radiotherapy had relapses but only 8.3% of patients who had received UKRAIN. During the following two years, 33.3% of patients who had received 5-FU and radiotherapy had relapses but only 16.7% of the patients who had been treated with UKRAIN (Bondar 1998). 90 patients with histologically verified advanced pancreatic cancer were treated in a controlled randomised study by Prof. Beger et al. at the University of Ulm, Germany. Patients in group A received gemcitabine 1000 mg/m2, those in group B received 20 mg UKRAIN, and those in group C received 1000 mg/m2 gemcitabine followed by 20 mg UKRAIN per week. Survival rates after six months were 26% in group A, 65% in group B and 74% in group C. The results showed that in inoperable advanced pancreatic cancer, survival rates were doubled using UKRAIN in combination with gemcitabine (Gansauge 2002). Patients were further observed after the conclusion of the study and it was noted that UKRAIN was well tolerated and could be administered without problem to all patients. UKRAIN brought about a significant increase in survival time in comparison to therapy with gemcitabine alone. Combination therapy with gemcitabine and UKRAIN showed no advantage over monotherapy with UKRAIN. The longest survival in the gemcitabine group was 19 months, 21 months in the gemcitabine+Ukrain group, and in the Ukrain group a patient was still alive after 28 months. The authors concluded: ‘As a result of this study we highly recommend the treatment of patients suffering from advanced pancreatic cancer with Ukrain’ (Gansauge 2003). The recent clinical study by the same group provided additional data on the benefit of a combined adjuvant treatment with Ukrain and gemcitabine. The relapse-free survival time was 21.7 months, and median survival 33.8 months which is clearly better than results reported in the literature earlier, for example 20.4 months in the study by Kurosaki et al, 2004. The actuarial survival rates were 86.7% after one year, 76.6% after two years, 46.7% after three years and 23.3% after five years (these patients are still alive). Again, this publication supports the efficacy (and safety) of the use of Ukrain as it demonstrates a considerable prolongation of survival compared to what is known from other clinical studies (Gansauge 2007). The selective effect of Ukrain as well as its mechanism of action has been proven in many studies by researchers from various countries. In the study by Hohenwarter et al. the inhibitory effect of Ukrain on malignant and normal cells was compared. To obtain a 50% growth inhibition, a tenfold concentration had to be used with normal endothelial cells compared to a human osteosarcoma cell line. Laser scanning microscopy showed a high uptake of Ukrain in malignant cells while the content in normal cells under the same experimental conditions was substantially lower (Hohenwarter et al, 1992; used cell lines: human osteosarcoma and melanoma cell lines; human endothelial cells from umbilical vein). At the 89th annual meeting of the American Association for Cancer Research in New Orleans, March 28 – April 1, 1998 A. Panzer and J.C. Seegers (University of Pretoria, South Africa) presented their work on the selective effect of Ukrain on various cancer cell lines. The authors concluded “that Ukrain is selectively toxic to malignant cells by causing a metaphase block which is characterised by abnormal chromosomal distribution, and results in the formation of micronuclei and in apoptosis” (Panzer and Seegers, 1998; used cell lines: human cervical carcinoma HeLa and human foreskin fibroblasts Hs27, squamous oesophageal cancer WHCO5; transformed human embryonic kidney Graham 293, and transformed African green monkey kidney Vero). In 2000 the group from South Africa published another article in which the selective mode of action of Ukrain was denied (Panzer et al, 2000). The authors did not explain the contradiction in the results of their two studies. The Ukrain opponents called the authors' group ‘independent’; however, the results of the study which denied the selective effect of Ukrain could not be reproduced by any other research group. In the tests on epidermoid carcinoma cells as well as prostate cancer cell lines, Roublevskaya et al. from Rochester University, USA revealed Ukrain to induce the G2M accumulation of cancer cells but not of normal cells. The researchers also observed an upregulation of CDK inhibitor p27 in cancer cell lines (Roublevskaya et al., 2000a, 2000b; used cell lines: epidermoid carcinoma ME180 and A431, prostate cancer LNCaP; normal human keratinocytes HaCaT). This malignocytolytic effect of Ukrain in prostate cancer was confirmed in clinic. 74 patients with prostatic carcinoma were involved in a controlled study at the Villa Medica Clinic (Edenkoben, Germany). These patients had already been treated according to all conventional therapy protocols, and due to relapse and/or progression of the disease had no further conventional options available. They were treated with UKRAIN, and partially at the same time with local hyperthermia, and the following results were obtained: complete remission (full tumor disappearence) in 73% of patients and partial remission (partial tumor disappearence) in 22%, with only 5% of patients showing no effect on the progress of the disease. Cordes et al. from Eberhard-Karls-University Tubingen, Germany, investigated the effects of Ukrain on cell survival, alteration of the cell cycle and induction of apoptosis without and in combination with ionising radiation (IR). Ukrain modulated radiation toxicity of human cancer cell lines and protected normal cells from radiation. The combination of Ukrain plus IR gave enhanced toxicity in CCL-221 and U-138MG cells but not in MDA-MB-231 and PA-TU-8902 cells. A radioprotective effect was found in normal human skin and lung fibroblasts (Cordes et al, 2002; used human cell lines: breast carcinoma MDA-MB-231, pancreas carcinoma PA-TU-8902, colorectal cancer CCL-221, glioblastoma U-138MG; skin fibroblasts HSF1, HSF2, and lung fibroblasts CCD32-LU). The mechanism of action of NSC 631570 on pancreas cancer was studied on human cell lines AsPC1, BxPC3, Capan1, MiaPaCa2 and Panc1. After pre-incubation, semiconfluent cultures were incubated with FCS-free medium for 24 hours and covered with FCS-supplemented medium and different concentrations of thiophosphoric acid, chelidonine and NSC-631570. Cells for staining procedures were cultured under similar conditions on chamber slides. For cell cycle analyses, proliferation assays and mitose phase analyses, cells were harvested 24 and 48 hours after incubation with 20 ng/ml, 60 ng/ml, 0.2 µg/ml, 0.6 µg/ml, 2 µg/ml, 6 µg/ml and 20 µg/ml chelidonine or 1 µg/ml, 5 µg/ml, 10 µg/ml and 20 µg/ml NSC-631570 or 0.144 µg/ml, 1.44 µg/ml and 2.88 µg/ml thiophosphoric acid or mixtures of chelidonine and thiophosphoric acid. Serial experiments at 4-hour intervals with AsPC1 and immunfluorescence stainings were performed using 0.6 µg/ml chelidonine or 10 µg/ml NSC-631570. Cell cycle analysis, proliferation assays, DNA laddering, Western blot analysis, and in vitro polymerisation of tubulin were then performed. Authors observed that NSC-631570 induces G2/M-arrest in pancreatic cancer cells. Furthermore, they discovered that it arrests cells in G2/M in a dose- and time-dependent fashion. Serial experiments also revealed that the effects on cell cycle phases caused by 10 µg/ml NSC-631570 are already irreversible after an incubation period of 8 hours. In-vitro polymerisation of monomeric tubulin revealed that NSC-631570 stabilizes tubulin monomers and as a result inhibits microtubule network assembly (Ramadani et al, 2000). In an experimental study assessing cell proliferation by BrdU-incorporation into pancreatic cancer cell lines (AsPC1, BxPC3, MiaPaCa2, Jurkat and THP-1) and cell cycle analysis by Giemsa staining, NSC 631570 (10 mg/ml) was shown to induce a high accumulation of treated cells in the G2/M phase after 24 hours incubation. The rate of apoptosis in similarly treated peripheral mononuclear cells did not show any differences between treated and untreated cells; further, mitogen-stimulated lymphocytes showed an increased blastogenic response (Gansauge et al, 2001). An Italian research group, Gagliano et al. from the University of Milan, used RT-PCR, Western blot and SDS-zymography to investigate the effects of Ukrain on the expression of genes and proteins involved in the extracellular matrix remodelling associated with tumour invasion in human cultured glioblastoma cells. There was a significant, dose-related decrease in glioblastoma cell proliferation and a tendency to the downregulation of SPARC (secreted protein acidic and rich in cysteine) after treatment with Ukrain, suggesting ‘the drug may be a useful therapeutic tool for brain tumours’ (Gagliano et al, 2006; used cell lines: human glioblastoma MI lines T60, T63, and GBM). Researchers at the Instituto Nacional de Cancerologia, Mexico City, Mexico ‘revealed that Ukrain induced apoptosis in a panel of cancer cell lines by activating the intrinsic cell death pathway. Interestingly, nontransformed fibroblasts (hTERT) cell line was insensitive to the drug.’ (Mendoza et al, 2006; used cell lines: cervical cancer HeLa, HeKB, HeKS32, HeBcl3, HeNFR and HeIKK, human colon cancer SW480, human renal carcinoma HEK293, human osteosarcoma MG-63, and immortalised human fibroblasts hTERT). The Italian research group has extended their research in glioblastoma. In the 2007 published study authors reported that after treatment with Ukrain, glial fibrillar acidic protein (GFAP) fluorescence increased and a higher number of glioblastoma cells displayed GFAP organized into a filamentous state. Western blot analysis confirmed that Ukrain tended to upregulate this protein. Connexin 43 was not modulated by Ukrain. Ukrain induced apoptotic rate was up to 28.9%, likely mediated by cytochrome c release in the cytoplasm (Gagliano et al, 2007). This selective effect of Ukrain on cancer cells has been proven not only with autofluorescence under UV-light (Nowicky et al, 1988) and pharmacokinetics (Doroshenko et al, 2000) but also in many in vitro experiments and several clinical studies. UKRAIN tests on healthy probands showed no side effects. At therapeutic dose Ukrain has minimal adverse effects. These adverse effects can occur in cancer patients during therapy and may be explained by the action of tumor degradation products. These effects disappear when the tumor is in complete remission. The biggest wish of all oncologists has been to develop an agent that is only toxic against cancer cells. For a long time this aim was considered impossible. The drug Ukrain makes the dream a reality and has taken the first big step towards the solution of this problem. The wide spectrum of cancer cell lines where Ukrain is effective explains its use in the treatment of various malignant tumours. In Ukraine, Georgia and Mexico Ukrain is officially registered for the treatment of various types of cancer. Thanks to the unique properties of the preparation the inventor of this medication was nominated for the Nobel Prize in 2005 and for the Alternative Nobel Prize in 2007. |
|